In the most simplest form, topoisomerases bind to the DNA to cut the phosphate backbone. Once the DNA is cut, it allows for the tension to be relieved or the knot to be released. After this occurs, the DNA backbone is resealed the the DNA is fixed.
(http://www.lookfordiagnosis.com/mesh_info.php?term=dna+topoisomerases%2C+type+i&lang=1)
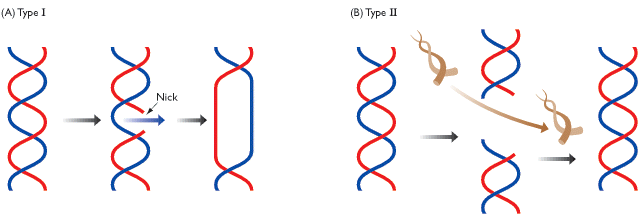
There are two different types of topoisomerases that function in the body. Type I (as you can see in the above diagram) just cuts through one strand of the DNA. This "nick" functions best at just relieving tension, but wouldn't help with knots that occur. This is where the type II topoisomerase comes in handy. Type II cuts through both strands of the DNA, allows for an unbroken piece of DNA to go through the break, and then finally reseals the strands. This fixes knots that would be expected to occur with the condensation of DNA.
Class II topoisomerases require ATP to fix knots. When it binds to one part of the DNA (G-segment), it uses ATP to bind to another parts (T-segment). The topoisomerase cuts the G-segments to allow the T-segment to pass through the break and fix the problem of the knot. Then the G-segment is glued back together and the topoisomerase releases. You can see this occur in the figure below.
(http://www.udel.edu/chem/bahnson/chem645/websites/Sapra/Mechanism%20of%20type%20II%20topoisomerase.html)
Because of its uses, topoisomerase inhibitors are increasingly being used as anti-cancer drugs. When these drugs interfere with enzyme activity, the topoisomerase may not be able to fix knots, or it will break the DNA, but not reseal the ends. Both of these could be detrimental to the cells. Replication would be inhibited which is a feature of cancer cells.
As you can expect, topoisomerase II are very important in your body for normal replication and transcription. These enzymes are fascinating in their way of releasing knots by introducing double-stranded breaks. Cell survival is impacted by the function of these enzymes and they deserve to be protein of the year.

(http://en.wikipedia.org/wiki/Type_II_topoisomerase)
Sources:
Champoux JJ (2001). "DNA topoisomerases: structure, function, and mechanism". Annu. Rev. Biochem. 70: 369–413.
http://www.ebi.ac.uk/interpro/potm/2006_1/Page2.htm
Willmore E, de Caux S, Sunter NJ et al. (2004). "A novel DNA-dependent protein kinase inhibitor, NU7026, potentiates the cytotoxicity of topoisomerase II poisons used in the treatment of leukemia". Blood 103 (12): 4659–65.
Great work on laying a foundation before diving into the specifics of this protein. I feel as if I understand what both topoisomerase class 1 and 2 does in the cell. Your pages all flow together to give a large amount of information in a way that many people can understand. But beyond cancer treatments what other diseases might topoisomerase class 2 help to treat?
ReplyDeleteGreat job on providing detailed but very clear information on the biochemistry of topoisomerases! I think it is very apparent from your blog posts just how important topoisomerases are in the body, as all biochemical processes depend on enzymes and other structures whose formation is dependent on transcription. Well done.
ReplyDeletenice and helpful blog
ReplyDelete